Abstract
Background
Withdrawal of life-sustaining measures is a common event in the intensive care unit yet it involves a complex balance of medical, legal and ethical considerations. Very few healthcare providers have been specifically trained to withdraw life-sustaining measures, and no comprehensive guidelines exist to help ensure clinicians deliver the highest quality of care to patients and families. Hence, we sought to develop guidelines for the process of withdrawing life-sustaining measures in the clinical setting.
Methods
We convened an interdisciplinary group of ICU care providers from the Canadian Critical Care Society and the Canadian Association of Critical Care Nurses, and used a modified Delphi process to answer key clinical and ethical questions identified in the literature.
Results
A total of 39 experienced clinicians completed the initial workshop, and 36 were involved in the subsequent Delphi rounds. The group developed a series of guidelines to address (1) preparing for withdrawal of life-sustaining measures; (2) assessment of distress; (3) pharmaceutical management of distress; and (4) discontinuation of life-sustaining measures and monitoring. The group achieved consensus on all aspects of the guidelines after the third Delphi round.
Conclusion
We present these guidelines to help physicians provide high-quality end of life (EOL) care in the ICU. Future studies should address their effectiveness from both critical care team and family perspectives.
Similar content being viewed by others
Introduction
Withdrawal of life-sustaining measures is a common event in critical care units in many parts of the world [1–3]. Withdrawal of life-sustaining measures is usually a planned and relatively brief procedure, but patients may experience distress due to dyspnea, pain, retention of airway secretions, and other symptoms. While physicians have an obligation to relieve these symptoms, they also have an obligation (in some countries) to avoid deliberately giving excessive symptom control medication at doses that might shorten life. The medical team must also prepare family members for what they will witness, anticipate any worries or concerns they may have, and support those who may wish to be present during the procedure. If withdrawal of life-sustaining measures is poorly handled it may cause distress for the patient, his or her family, and the entire ICU team.
Critical care societies have published recommendations for end of life (EOL) care [4, 5], and some authors have published clinical recommendations for withdrawal of life-sustaining measures [6–9] (Table 1). Yet there is considerable variation in practice about how life-sustaining measures are withdrawn [2, 10]. A recent review of EOL care in the ICU by Cook and Rocker concluded that “there is no single, universally-accepted technical approach” [11]. Furthermore, some reports have highlighted unclear or inconsistent orders [12–14], which may lead to confusion, uncertainty, and distress.
Our goal was to develop practice guidelines on behalf of the Canadian Critical Care Society (CCCS) for the process of withdrawing life-sustaining measures in order to improve the quality of EOL care provided in intensive care units. To achieve this goal we convened a meeting of a large interprofessional group of experienced ICU care providers, and used a Delphi process to achieve consensus on important clinical questions.
Methods
Design
Modified Delphi process.
Participants
We convened an interdisciplinary group of ICU providers from the CCCS, the Canadian Association of Critical Care Nurses, as well as ICU-based respiratory therapists and pharmacists partnered with these organizations. Potential participants were identified purposively to represent different geographic regions and clinical practices.
Literature review
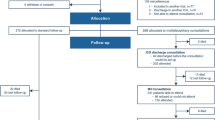
In order to frame and inform the work of the working group, two authors (JD and JWD) conducted a literature search to identify articles relevant to the process of withdrawing life-sustaining measures (see Electronic Supplementary Material Appendix 1). Keywords were identified from relevant articles, and these keywords were used to perform a search of MEDLINE (1946 to October week 1, 2013), Embase (1974–2013 October 10), and EBM Reviews (to 2013) that identified 1983 articles (Fig. 1). After duplicate screening of the articles, 53 were found to be relevant. A search of the references for these articles identified an additional 46 articles that were included in the review. The full methods and results of this review will be published separately. A qualitative synthesis identified four broad categories of clinical considerations: (1) preparing for withdrawal of life-sustaining measures; (2) assessment of distress; (3) pharmaceutical management of distress; and (4) discontinuation of life-sustaining measures and monitoring. A fifth category of considerations, bereavement support, was combined with category 1 for the purpose of guideline development. Two of the authors (JD and JWD) then reframed these considerations as statements (e.g. “Opioids and sedatives should be titrated to symptoms with no specified dose limit during withdrawal of life-sustaining measures”) that could be considered and evaluated by the participants during the Delphi process. Note that these articles and the subsequent questions were intended to catalyze and inform the discussions of the working group, not to constrain the guidelines in any way.
Grading system
After reviewing the literature, the authors found that there was very little evidence to support specific practices. Only four of the 99 articles involved an intervention, and only one of these was randomized—the remainder were observational or qualitative studies, reviews, guidelines, or opinionated editorials. In addition, we recognized that some recommendations would be framed by legal or ethical considerations, regardless of published evidence or opinion. Accordingly, we chose not to use the GRADE system for recommendations [25], since virtually every recommendation would need to be weak given the quality of evidence; the uncertainty about the balance between desirable and undesirable effects; the variability in values and preferences; and the lack of data about costs and resource use. As a consequence we developed a unique grading system that would allow participants to integrate the limited evidence with their own experience and ethical/legal considerations to assign one of three levels of recommendation:
-
1.
We recommend Multiple studies indicate a need for this action, and it is acceptable within current legal and ethical frameworks.
-
2.
We suggest The need for this action is supported by a single study or multiple expert opinions, and it is acceptable within current legal and ethical frameworks.
-
3.
No recommendation The need for this action is not supported by studies or multiple expert opinions and/or it is legally or ethically questionable. In practice, this level of recommendation was given only to statements with insufficient support in the literature; those that were considered legally or ethically questionable were removed entirely after round 1.
This grading system was accepted by all participants. Subsequently, two authors (JD and JWD) assigned initial levels of recommendation to each of the initial statements based purely on the evidence identified in the literature review. These are provided in the Electronic Supplementary Material Appendix 2.
Delphi process
All participants attended an introductory session at the Canadian Critical Care Forum in November 2013 that included a review of the literature and an expert overview of the legal and ethical framework in Canada. Participants were divided into four prespecified subgroups, each tasked with addressing one of the categories of clinical consideration identified above. Each subgroup consisted of a balance of experienced physician and nursing representation from across the country. Pharmacy representatives were assigned to the group addressing pharmacologic management of distress, and respiratory therapists were assigned to the group addressing the process of withdrawal of life-sustaining therapies.
Each subgroup was provided with copies of the literature review and original articles to inform their discussion. Two subgroups required subsequent meetings, which took place over the spring and summer of 2014 by teleconference, to complete the first round. Subsequent rounds took place using an online survey tool (SurveyMonkey™).
In round 1, each subgroup was instructed to begin their discussion by reviewing their list of statements generated from the literature, as well as additional statements identified by the authors (see Electronic Supplementary Material Appendix 2). Subgroups were asked to add, delete, or modify statements for clarity (these revised statements appear in the Electronic Supplementary Material Appendix 2), and then vote openly to assign a level of recommendation to each statement. The consensus opinion for the initial recommendation level of the subgroup was set a priori to be the highest level of recommendation supported by a simple majority (i.e. 50 %) of the subgroup members.
In round 2, we circulated the full list of clinical statements to the entire working group by online survey, and asked each member to anonymously assign their level of recommendation to each question. Participants were informed of the level of recommendation assigned by the subgroup in round 1. Again, the consensus opinion for the initial recommendation level of the group was set a priori to be the highest level of recommendation supported by at least 50 % of the group members.
In round 3, we assembled the individual statements into paragraphs organized by theme, and subdivided by level of recommendation assigned in round 2. In this round, the goal was to determine the level of agreement of the participants with the paragraphs generated (both the content and the level of recommendation). Accordingly, we sent these paragraphs to all participants and asked them to rate their agreement with each paragraph on a 5-point Likert scale. We predefined a score of 4 or 5 as “agreement”, and set a higher threshold of 70 % agreement for acceptance of each paragraph. We have previously used this higher threshold for consensus (rather than the simple majority used to assign the initial recommendation level) when considering statements that might be ethically contentious [26]. Participants who assigned a score of 3 or less were asked to suggest changes to the paragraph. Since we achieved consensus for all of the paragraphs during round 3, subsequent rounds were not required. The final guidelines underwent minor revisions for clarity of language or to remove duplication.
Results
In total, we had 39 participants in the original workshop (one withdrew during the workshop), and 36 participants agreed to participate in subsequent Delphi rounds. Of the original participants, 28 were experienced critical care physicians representing CCCS members, eight were experienced representative of the Canadian Association of Critical Care Nurses, two were experienced pharmacists affiliated with the CCCS, and two were experienced respiratory therapists affiliated with the CCCS. We had a response rate of 30/36 (83 %) in the second Delphi round and 32/36 (89 %) in the third Delphi round. The working group accepted all paragraphs in the third round.
In rounds 1 and 2, the participants often assigned a level of recommendation that was stronger than would have been warranted by the evidence base (see Electronic Supplementary Material Appendix 2). This led to many statements being given the highest level of recommendation (“Recommend”), whereas the evidence only justified this level of recommendation for one statement. Table 2 summarizes the major areas of discussion for each subgroup. In round 3, the level of acceptance was very high for most paragraphs (Table 3 provides an executive summary of the final recommendations; Table 4 lists the results of round 3), but particularly for Section 1 (preparing for withdrawal of life-sustaining measures, >90 %) and Section 4 (discontinuation of treatment and monitoring, >87 %). The paragraphs with the lowest level of acceptance were in Section 3 (pharmaceutical management of distress, >77 %). In this section, respondents who disagreed with the paragraphs generally felt that the guidelines were too specific and prescriptive.
Section 1: preparing for withdrawal of life-sustaining measures
1.1 Informing the patient/family members
We RECOMMEND that the interdisciplinary team assess each patient’s desire to discuss withdrawal of life-sustaining measures and respond accordingly. Patients (when appropriate) and family members should be informed about the process; the role of each team member; and what to expect during the dying process (e.g. changes in appearance and vital signs); physical signs of distress that they may witness; and how distress will be assessed and treated. Family members should be informed that the time between withdrawal of life-sustaining measures and death may be variable and difficult to predict. All members of the team play an important role in educating and supporting patients and families during withdrawal of life-sustaining measures. Hospital spiritual care and/or social work should be invited to attend discussions of withdrawal of life-sustaining measures. Young children of adult patients should receive particular attention and support from the team.
We SUGGEST that members of the interdisciplinary team be involved in discussions with the patient/family about what to expect during withdrawal of life-sustaining measures.
1.2 Team preparation and availability
We RECOMMEND that an interprofessional care plan for withdrawal of life-sustaining measures be outlined for each patient, and that an experienced physician, nurse, and respiratory therapist be readily available during withdrawal of life-sustaining measures to respond quickly to significant signs of distress at the time of extubation, or to other symptoms that arise. These three team members should be present at the bedside for cases that are anticipated to be difficult, such as patients who are awake; those receiving large doses of comfort medications at baseline; those with a high respiratory drive; and cases where the family or team are likely to become highly emotional.
We SUGGEST that physicians return to the bedside at regular intervals to ensure emotional and psychological support of patients/families if appropriate.
1.3 Setting
We RECOMMEND that whenever possible, patients should be moved to a private room, visitation should be liberalized, and monitors should be turned off within the room to minimize distraction. The team should use an unobtrusive signal to the rest of the ICU (e.g. a sign on the door or curtain) that a withdrawal of life-sustaining measures is in progress, in order to ensure a respectful and quiet atmosphere around the room. Family members should be offered a private quiet room separate from the patient’s room (when possible).
1.4 Interdisciplinary communication
We RECOMMEND that physicians and nurses should communicate clearly about the patient’s comfort level, and both should participate in providing palliative care.
1.5 Debriefing
We RECOMMEND that the interprofessional team consider formal debriefing sessions following difficult cases of withdrawal of life-sustaining measures, when the team experiences intense emotion, or when new staff members are involved.
1.6 Grief and bereavement support
We RECOMMEND that family members be offered acute grief support at the time of withdrawal of life-sustaining measures and death, and offered a referral to community bereavement support services. Family members should also be sent a letter of condolence [27] that includes bereavement support information. ICUs should compile a list of resources for grief and bereavement support, including support groups in their area.
We SUGGEST that the interprofessional team be educated on acute bereavement support for grieving family members and friends. Family members should be educated about the grieving process, and offered a future opportunity to debrief with the physician and interprofessional team. A member of the interprofessional team should contact bereaved family members a few weeks or months after the patient’s death to answer any questions and assess coping.
1.7 Involving family members in patient care
We RECOMMEND that family members be allowed to be present at the bedside during withdrawal of life-sustaining measures, and that they be allowed to participate in patient care as appropriate. Family members should be encouraged to “be” with their loved ones and celebrate the life of the patient through memory books, expressions of life, cultural or religious rituals, and other specific ways to celebrate the uniqueness of the patient. Cultural or religious rituals should be facilitated when possible, and family members should be allowed to involve their own clergy or spiritual support. Family members should be allowed to stay overnight in the ICU.
Section 2: assessment of distress
2.1 Assessment of pain
We RECOMMEND that pain be assessed using a standardized scoring system, such as the Behavioural Pain Scale (BPS) or the Critical Care Pain Observation Tool (CCPOT). Family members should be involved in decisions about whether the patient is in pain.
We SUGGEST that pain assessments can be based on objective signs such as tachypnoea, tachycardia, diaphoresis, accessory muscle use, nasal flaring, grimacing, rigidity, wincing, shutting of eyes, clenching of fists, verbalizing, and moaning.
We offer NO RECOMMENDATION about the use of the Pain Assessment Behaviour Scale (PABS), the use of specific vital sign thresholds as indicators of pain (e.g. HR >100, RR >35), or the use of an interprofessional team of raters.
2.2 Assessment of agitation
We RECOMMEND that agitation be assessed using a standardized assessment scale that is validated in critically ill patients, such as the Sedation-Agitation Score (SAS) or the Ramsay Agitation Sedation Scale (RASS).
We offer NO RECOMMENDATION about the use of bispectral EEG monitoring to assess agitation during withdrawal of life-sustaining measures.
2.3 Assessment of respiratory distress
We SUGGEST the use of a standardized assessment tool such as the Respiratory Distress Observation Scale (RDOS) to assess respiratory distress. The presence of tachypnoea, a rising respiratory rate (>50 % from baseline), tachycardia, a fearful facial expression, accessory muscle use, paradoxical breathing, or nasal flaring can be interpreted as respiratory distress. Family members should be involved in decisions about whether the patient is in respiratory distress.
We offer NO RECOMMENDATION about the use of a specific respiratory rate threshold as an indicator of respiratory distress (e.g. >35 breaths per minute), or the use of an interdisciplinary team of raters to assess respiratory distress.
2.4 Assessment of delirium
We RECOMMEND that, when appropriate, patients should be assessed for delirium during withdrawal of life-sustaining measures using a standardized assessment tool that is validated in critically ill patients (e.g. CAM-ICU, ICDSC).
2.5 Documentation of treatment of pain and distress
We RECOMMEND that whenever a patient is given any medication to treat or prevent symptoms during withdrawal of life-sustaining measures, the rationale should be documented using the criteria specified in the orders (e.g. “for accessory muscle use”).
Section 3: pharmaceutical management of distress
3.1 The goal of pharmaceutical management
We RECOMMEND that pharmaceutical agents be used to treat symptoms, and also to PREVENT symptoms when they may be anticipated but are not yet apparent.
3.2 Neuromuscular blockade
We SUGGEST that paralytic medications be discontinued and their effects allowed to wear off enough so that the team can appreciate signs of distress prior to withdrawal of life-sustaining measures (Train-of-four ≥3/4).Footnote 1
3.3 Choice of opioid and sedative medications
We RECOMMEND that if the patient is already comfortable on a stable dose of opioid and/or sedative, these should be continued at those doses during WDLS. For opioid-naïve patients, morphine is the initial opioid of choice for pain or dyspnoea during withdrawal of life-sustaining measures.
We SUGGEST that sedatives such as barbiturates or propofol should be used as second-line for sedation during withdrawal of life-sustaining measures, when benzodiazepines are ineffective or in exceptional circumstances.
3.4 Titration of opioids
We RECOMMEND that opioids should be titrated to symptoms with no specified dose limit during withdrawal of life-sustaining measures. In symptomatic, opioid-naïve adults, the starting bolus dose of intravenous morphine is 2 mg (or equianalgesic dose of another opioid), titrated to effect. The starting dose can be adjusted on the basis of considerations of size, age, and organ dysfunction. If a patient is receiving an infusion of morphine or hydromorphone, and he/she develops pain or respiratory distress, it is reasonable to give a bolus dose of two times the hourly infusion dose. Intravenous morphine/hydromorphone bolus doses should be ordered every 15 min as required, and intravenous fentanyl bolus doses should be ordered every 5 min as required. If a patient receives two bolus doses in an hour, it is reasonable to double the infusion rate.
We SUGGEST that pain or respiratory distress should be treated with an IV bolus dose or an opioid followed by a continuous opioid infusion.
3.5 Titration of sedatives
We RECOMMEND that sedatives should only be used once pain and dyspnoea are effectively treated with opioids, but combinations of opioids and sedatives can be used for symptom management during withdrawal of life-sustaining measures. Sedatives should be titrated to symptoms with no specified dose limit during withdrawal of life-sustaining measures. For sedative-naïve patients, symptoms should be treated with an intravenous bolus dose of sedative followed by an infusion. In symptomatic patients who are benzodiazepine-naïve it is reasonable to give a 2 mg intravenous bolus dose of midazolam followed by an infusion of 1 mg/h. These doses can be adjusted on the basis of considerations of size, age, and organ dysfunction. If a patient is receiving an infusion of midazolam and becomes symptomatic, it is reasonable to give a bolus dose equal to or double the hourly infusion dose. Intravenous midazolam bolus doses should be ordered every 5 min as required. If a patient receives two bolus doses in an hour, it is reasonable to double the infusion rate. Propofol is an alternative sedative to midazolam for patients who are already comfortable on a stable infusion of propofol, or for physicians who are familiar with using propofol as a sedative during withdrawal of life-sustaining measures.
3.6 Other medications
We SUGGEST that inhaled epinephrine should be used to treat post-extubation stridor in the conscious patient. Antinauseant medications should be ordered pro re nata (PRN) with opioids.
We offer NO RECOMMENDATION about the routine use of anticholinergic medication (e.g. butylscopolamine) pre-extubation to prevent upper airway secretions; the routine use of furosemide to prevent congestive heart failure; and the routine use of methylprednisolone to prevent post-extubation stridor.
Section 4: discontinuation of treatment and monitoring
4.1 The use of protocols
We SUGGEST that each ICU should develop and utilize protocols for withdrawal of mechanical ventilation and life-sustaining treatments.
4.2 Discontinuation of treatment
We RECOMMEND that all non-comfort medications, blood transfusions, haemodialysis, vasopressors, inotropes, parenteral nutrition, enteral tube feeding, antibiotics, intravenous fluids, and blood work be discontinued.
We SUGGEST that supplemental oxygen should not be provided unless it is needed for comfort.
4.3 Order and pace of discontinuation
We RECOMMEND that the pace of withdrawal of life-sustaining measures be individualized for each patient. Typically, vasopressors and inotropes should be discontinued first, followed by mechanical ventilation and any artificial airway in a stepwise manner. Life-sustaining treatments should be withdrawn in a stepwise manner, ensuring alleviation of any pain, respiratory distress, or anxiety at each step.
4.4 Discontinuation of ventilation
We RECOMMEND that the sequence and process of withdrawal of mechanical ventilation must be individualized with comfort as the paramount goal. Mechanical ventilation should be withdrawn as quickly as possible, with the speed determined by the time it takes to achieve comfort at each step. Patients should not be routinely extubated to non-invasive mechanical ventilation. Pharmacologically paralysed patients should typically not be extubated or weaned to a spontaneous ventilator mode, continuous positive airway pressure, or a T-piece.
We SUGGEST that in most cases, the goal should be to extubate patients to room air. Extubation is preferable to leaving the patient intubated on minimal or no mechanical ventilatory support, but either option is acceptable.
4.5 Discontinuation of monitoring
We RECOMMEND that all monitoring that is not aimed at achieving comfort should be discontinued.
Discussion
We used a modified Delphi technique with a multidisciplinary expert panel to develop guidelines for quality EOL care during withdrawal of life-sustaining measures. These guidelines are intended to help clinicians provide high-quality EOL care by balancing the medical, legal, and ethical considerations that arise during withdrawal of life-sustaining measures in a manner suggested by a group of experienced professionals. These guidelines are not evidence-based; they were developed to provide a resource for front-line clinicians who are planning to withdrawal of life-sustaining measures and would like to benefit from evidence-informed consensus opinions from an interdisciplinary panel of experienced professionals in the field.
Previous reports have highlighted practices that drew concern from members of healthcare teams, and at least one episode that prompted an investigation by the US Office of the Inspector General [14]. One of the recommendations from this investigation was to develop comfort care guidelines and an order set, among other things. Treece et al. reported high nurse and physician satisfaction associated with the development of a withdrawal of life-sustaining measures protocol, although there was no reported change in the quality of death and dying according to bedside nurses [28]. This highlights the need to study the effect of implementing these guidelines, in order to determine whether they actually result in improvements in patient care.
These guidelines may alleviate concerns about the process of WDLS for organ donation after cardiac death. Previous reports have identified concerning cases of organ donation in which physicians were suspected of managing withdrawal of life-sustaining measures differently from normal practice in the hope of optimizing donor organ function [29]. Although not specific to organ donation, these guidelines can be used to ensure that consistency of EOL care is maintained. If physicians manage all cases of WDLS using the same management principles, regardless of whether or not DCD is considered, this will hopefully eliminate any concerns and perception that organ function might be given priority over the management of the donor.
These guidelines focus on the practical aspects of withdrawing life-sustaining therapies from patients whose treatment goals are palliative and who are not expected to survive long after WDLS. The discussion and framework for deciding whether or not to WDLS is beyond the scope of this project, and the general assumption of these guidelines is that the decision to WDLS has involved the patient or family members to a degree consistent with ethical and legal requirements. The CCCS position paper on withholding or withdrawal of life support [30] remains a relevant and useful resource in Canada, and others are available to inform practitioners in other jurisdictions [5]. In addition, regulatory bodies in most jurisdictions have established policies to guide physician behaviour in this area. These WDLS guidelines are meant to supplement other important aspects of EOL care that have been comprehensively reviewed by Rocker and Cook [11] to provide the highest-quality EOL care for patients dying in the ICU.
Because WDLS guidelines must integrate medical, ethical, and legal considerations, these guidelines may not apply to jurisdictions with different ethical and legal norms. For example, in many parts of the world, WDLS itself is illegal or strongly avoided [2, 3, 15, 31]. Our guidelines suggest a preference for extubation over leaving patients intubated; some practitioners may have a stronger preference for a “soft landing”—a slow WDLS without sudden events that might be perceived as precipitating death (e.g. extubation) [32]. Some patients and staff do not consider feeding and fluids to be medical therapies, and they would not consider it acceptable to withhold these even during WDLS [33]. It would be unrealistic to expect that one set of guidelines would be universally relevant and acceptable. But these guidelines might prompt other jurisdictions to develop their own WDLS guidelines, also with the intention of promoting good medical practice.
These guidelines are also intended for a general critical care population and reflect limitations in our ability to detect distress in some critically ill patients. For example, most of the stronger recommendations in Section 2 (“Assessment of distress”) involve assessments of distress based on patient behaviours, but these behaviours may not be present in patients with brain injury. At the same time, some patients in a vegetative state may not be able to experience pain or anxiety, in which case they would not require symptomatic treatment [9]. These factors may explain why neurologically injured patients tend receive lower doses of opioids (and sometimes no opioids at all) during WDLS [34, 35], and why clinicians may approach WDLS differently in this population. In addition, the guidelines state that chemically paralysed patients can undergo WDLS even before the paralysis has worn off, albeit in exceptional cases and with some modifications in approach (see Electronic Supplementary Material Appendix 3). Some would consider this to be a controversial recommendation that would require extensive documentation and justification in the patient’s chart, and may even be illegal in some countries.
The degree of consensus was quite high for most sections of the guidelines, but some working group participants expressed concern that Section 3 (“Pharmaceutical management of distress”) was too specific and prescriptive. For example, the guidelines suggest that a patient requiring more than one bolus dose of opioid for pain or dyspnoea should be started on an infusion of opioids. This may represent a desire of working group members to avoid the risk of undertreating symptoms in a patient who is expected to die, even if this means that the patient might receive more opioid than necessary.
Strengths of these guidelines include the use of a large, multiprofessional group of experts, and an iterative consensus process to ensure that they reflect well-considered opinions, informed by the evidence. Overall, there was a high degree of consensus for all aspects of the guidelines despite reports of variable practices [12–14], a poor evidence base, uncertainty about the balance between desirable and undesirable outcomes, lack of cost and resource data, and the controversial nature of WDLS. These factors led us to develop a new system of recommendations rather than use the GRADE system.
Limitations of these guidelines include the fact that our expert group was all from one country and included a purposively selected group of 36 experts. Furthermore, the quality of the evidence is low; most of the literature consists of expert opinion and observational studies. For example, one section (2.3) suggested the use of the Respiratory Distress Observation Scale (RDOS), which has not been validated in this setting. For this reason, we used a recommendation system that integrated both ethical and clinical considerations. Notably, the fact that so many of the guidelines were given a high strength of recommendation suggests that many participants attached a high value to low-quality evidence, or gave a high recommendation despite the lack of evidence. Although we emphasized the instructions regarding the strength of recommendation, we did not enforce the evidence criterion, nor did we restrict the guidelines to issues raised in the literature. We felt that it was more important to respect a strong recommendation from experts than to limit recommendations on the basis of a poor evidence base. Finally, although we included participants from non-medical professions, we did not have any patient advocates or family participants, or members of the clergy. Clearly, high-quality EOL care involves a respect for patient wishes whenever possible, and an incorporation of family preferences to the degree possible. The guidelines show the strong deference given to patient and family perspectives in Section 1.
We recognize that our guidelines were also developed in the context of a changing legal and ethical landscape with regards to Physician-Hastened Death (PHD). At the time of guideline development, PHD was not legal in Canada. The Supreme Court of Canada did strike down the legal prohibition of PHD in February 2015 [36], but since this decision only applies to adults capable of providing explicit consent, this decision is unlikely to affect the care provided to most patients in Canadian ICUs, and does not apply outside of Canada. PHD may be considered acceptable for incapable patients in ICUs in other countries [37, 38].
In conclusion, we present these guidelines for WDLS to help physicians provide high-quality EOL care in the ICU. Future studies should address their effectiveness from both critical care team and family perspectives. More research is needed to expand the evidence base in this area.
Notes
An additional recommendation regarding neuromuscular blockade has been removed from these guidelines but appears in an online supplement.
References
Prendergast TJ, Claessens MT, Luce JM (1998) A national survey of end-of-life care for critically ill patients. Am J Respir Crit Care Med 158(4):1163–1167
Mark NM, Rayner SG, Lee NJ, Curtis JR (2015) Global variability in withholding and withdrawal of life-sustaining treatment in the intensive care unit: a systematic review. Intensive Care Med 41(9):1572–1585
Sprung CL, Cohen SL, Sjokvist P et al (2003) End-of-life practices in European intensive care units: the Ethicus Study. JAMA 290(6):790–797
Lanken PN, Terry PB, DeLisser HM et al (2008) An official American Thoracic Society clinical policy statement: palliative care for patients with respiratory diseases and critical illnesses. Am J Respir Crit Care Med 177(8):912–927
Truog RD, Campbell ML, Curtis JR et al (2008) Recommendations for end-of-life care in the intensive care unit: a consensus statement by the American Academy of Critical Care Medicine. Crit Care Med 36(3):953–963
Rubenfeld GD (2004) Principles and practice of withdrawing life-sustaining treatments. Crit Care Clin 20(3):435–451 (ix)
Brody H, Campbell ML, Faber-Langendoen K, Ogle KS (1997) Withdrawing intensive life-sustaining treatment—recommendations for compassionate clinical management. N Engl J Med 336(9):652–657
Campbell ML (2004) Terminal dyspnea and respiratory distress. Crit Care Clin 20(3):403–417 (viii–ix)
Hawryluck LA, Harvey WR, Lemieux-Charles L, Singer PA (2002) Consensus guidelines on analgesia and sedation in dying intensive care unit patients. BMC Med Ethics 3:E3
Hall RI, Rocker GM (2000) End-of-life care in the ICU: treatments provided when life support was or was not withdrawn. Chest 118(5):1424–1430
Cook D, Rocker G (2014) Dying with dignity in the intensive care unit. N Engl J Med 370(26):2506–2514
Hunter JK, Dean T, Gowan J (2006) Death with dignity: devising a withdrawal of treatment process. Br J Nurs 15(3):138–140
Keenan SP, Mawdsley C, Plotkin D, Sibbald WJ (2003) Interhospital variability in satisfaction with withdrawal of life support: room for improvement? Crit Care Med 31(2):626–631
Kuschner WG, Gruenewald DA, Clum N, Beal A, Ezeji-Okoye SC (2009) Implementation of ICU palliative care guidelines and procedures: a quality improvement initiative following an investigation of alleged euthanasia. Chest 135(1):26–32
Paruk F, Kissoon N, Hartog CS et al (2014) The Durban world congress ethics round table conference report: III. Withdrawing mechanical ventilation—the approach should be individualized. J Crit Care 29(6):902–907
von Gunten C, Weissman DE (2003) Information for patients and families about ventilator withdrawal. J Palliat Med 6(5):775–776
von Gunten C, Weissman DE (2003) Ventilator withdrawal protocol. J Palliat Med 6(5):773–774
Von Gunten C, Weissman DE (2003) Symptom control for ventilator withdrawal in the dying patient. J Palliat Med 6(5):774–775
Truog RD, Cist AF, Brackett SE et al (2001) Recommendations for end-of-life care in the intensive care unit: The ethics committee of the Society of Critical Care Medicine. Crit Care Med 29(12):2332–2348
Marr L, Weissman DE (2004) Withdrawal of ventilatory support from the dying adult patient. J Support Oncol 2(3):283–288
Cist AF, Truog RD, Brackett SE, Hurford WE (2001) Practical guidelines on the withdrawal of life-sustaining therapies. Int Anesthesiol Clin 39(3):87–102
Kompanje EJ, van der Hoven B, Bakker J (2008) Anticipation of distress after discontinuation of mechanical ventilation in the ICU at the end of life. Intensive Care Med 34(9):1593–1599
Eschun GM, Jacobsohn E, Roberts D, Sneiderman B (1999) Ethical and practical considerations of withdrawal of treatment in the intensive care unit. Can J Anaesth 46(5 Pt 1):497–504
Campbell ML (2007) How to withdraw mechanical ventilation: a systematic review of the literature. AACN Adv Crit Care 18(4):397–403 (quiz 344–395)
Guyatt GH, Oxman AD, Vist GE et al (2008) GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ 336(7650):924–926
Downar J, Hawryluck L (2010) What should we say when discussing “code status” and life support with a patient? A Delphi analysis. J Palliat Med 13(2):185–195
Bedell SE, Cadenhead K, Graboys TB (2001) The doctor’s letter of condolence. N Engl J Med 344(15):1162–1164
Treece PD, Engelberg RA, Crowley L et al (2004) Evaluation of a standardized order form for the withdrawal of life support in the intensive care unit. Crit Care Med 32(5):1141–1148
McKinley J (2008) Surgeon accused of speeding a death to get organs. NY Times
Rocker G, Dunbar S (2000) Withholding or withdrawal of life support: the Canadian Critical Care Society position paper. J Palliat Care 16(Suppl):S53–S62
Levin PD, Sprung CL (2005) Withdrawing and withholding life-sustaining therapies are not the same. Crit Care 9(3):230–232
Aita K, Kai I (2010) Physicians’ psychosocial barriers to different modes of withdrawal of life support in critical care: a qualitative study in Japan. Soc Sci Med 70(4):616–622
Bulow HH, Sprung CL, Reinhart K et al (2008) The world’s major religions’ points of view on end-of-life decisions in the intensive care unit. Intensive Care Med 34(3):423–430
Mayer S, Kossoff SB (1999) Withdrawal of life support in the neurological intensive care unit. Neurology 52(8):1602–1609
Mazer MA, Alligood CM, Wu Q (2011) The infusion of opioids during terminal withdrawal of mechanical ventilation in the medical intensive care unit. J Pain Symptom Manag 42(1):44–51
Carter v. Canada (Attorney General), 5 (Supreme Court of Canada 2015)
Kompanje EJ, Epker JL, Bakker J (2014) Hastening death due to administration of sedatives and opioids after withdrawal of life-sustaining measures: even in the absence of discomfort? J Crit Care 29(3):455–456
Vincent JL, Schetz M, De Waele JJ et al (2014) “Piece” of mind: end of life in the intensive care unit statement of the Belgian Society of Intensive Care Medicine. J Crit Care 29(1):174–175
Acknowledgments
The authors wish to thank Drs. Tasnim Sinuff (University of Toronto, Toronto) and John Muscedere (Queen’s University, Kingston) for methodological advice during the guideline development process. We wish to thank Tracey Bailey (John Dossetor Health Ethics Centre and Department of Psychiatry, Faculty of Medicine and Dentistry, University of Alberta, Edmonton) and Dr. Natalie Bandrauk (Faculty of Medicine, Memorial University, St. John’s) for providing an expert overview of the legal and ethical framework of WDLS in Canada for the workshop participants. We also wish to thank the Delphi participants for providing their expert opinions.
The authors would also like to thank all of the working group members: Margaret Herridge (University of Toronto, Toronto), Tereza Coughlin (University of Alberta, Edmonton), Linda Nusdorfer (Sunnybrook Health Sciences Centre, Toronto), Kim Bowman (Trillium Gift of Life, Toronto), Shavaun MacDonald (University of Alberta, Edmonton), Mary O’Brien (Memorial University, Newfoundland), Adrian Robertson (University of Manitoba, Winnipeg), Tobias Witter (Dalhousie University, Halifax), Catherine Farrell (Universite de Montreal, Montreal), Angie Payne (Eastern Health, Newfoundland and Labrador), Lisa Burry (Mount Sinai Hospital, Toronto), Richard Hall (Dalhousie University, Halifax), Karen Dryden-Palmer (Hospital for Sick Children, Toronto), Kavita Sridhar (Lakeridge Health, Oshawa), Alexis Turgeon (Universite de Laval, Quebec), David Zygun (University of Alberta, Edmonton), Bojan Paunovic (University of Manitoba, Winnipeg), Peter Cox (Hospital for Sick Children, Toronto), Ruth Trinier (Hospital for Sick Children, Toronto), Claudio Martin (University of Western Ontario, London), Gordon Rubenfeld (University of Toronto, Toronto), Kirk Dawe (Eastern Health, Newfoundland and Labrador), Alison Fox–Robichaud (McMaster University, Hamilton), Sam Minor (Dalhousie University, Halifax), Eli Malus (Windsor Regional Hospital, Windsor), Kim Wiebe (University of Manitoba, Winnipeg), Marie Edwards (University of Manitoba, Winnipeg), Natalie Bandrauk (Memorial University, Newfoundland), Viriginie Bibeau (Universite de Laval, Quebec), John Granton (University of Toronto, Toronto), Michael Sharpe (University of Western Ontario, London), William Gallacher (Dalhousie University, Halifax), Anand Kumar (University of Manitoba, Winnipeg), Karen Choong (McMaster University, Hamilton), Joann Kawchuk (University of Saskatchewan, Saskatoon), Douglas Matheson (University of Alberta, Edmonton), Brenda Morgan (University of Western Ontario, London), Tara Fowler (University Health Network, Toronto), Angela Mcgauley (University Health Network, Toronto).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None of the authors have any conflicts of interest to declare that are relevant to this manuscript.
Additional information
On behalf of the Canadian Critical Care Society’s Withdrawal of Life-Sustaining Treatment Working Group.
Take-home message: We used a Delphi process and an interprofessional group of experienced clinicians to develop guidelines for the process of withdrawal of life-sustaining measures (WDLS) in the clinical setting. These guidelines can help clinicians provide high-quality EOL care by balancing the medical, legal, and ethical considerations that arise during WDLS.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Downar, J., Delaney, J.W., Hawryluck, L. et al. Guidelines for the withdrawal of life-sustaining measures. Intensive Care Med 42, 1003–1017 (2016). https://doi.org/10.1007/s00134-016-4330-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-016-4330-7