Abstract
Objective
To assess the prevalence and the impact of chronic and/or acute non-respiratory co-morbidity on short and longer-term outcome of non-invasive positive pressure ventilation (NIPPV) in acute decompensations of chronic obstructive pulmonary disease (COPD) with acute hypercapnic respiratory failure (AHRF).
Design and setting
An observational study in a three-bed respiratory monitoring unit in a respiratory ward of a non-university hospital.
Patients
We grouped 120 consecutive COPD patients requiring NIPPV for AHRF (pH 7.28±0.05, PaO2/FIO2 ratio 192±63, PaCO2 78.3±12.3 mmHg) according to whether NIPPV succeeded (n=98) or failed (n=22) in avoiding the need for endotracheal intubation and whether alive (n=77) or dead (n=42) at 6 months.
Measurements and results
The prevalence of chronic and acute co-morbidity was, respectively, 20% and 41.7%; most of the cases were cardiovascular. In-hospital NIPPV failure was greater in patients with than in those without chronic (33.3% vs. 14.6%) or acute co-morbidity (32% vs. 8.6%). Six-month mortality was worse in patients with than in those without chronic (54.2% vs. 30.5%) or more than one acute co-morbidity (66.7% vs. 30.8%). Multiple regression analysis predicted in-hospital NIPPV failure by acute co-morbidity and forced expiratory volume in 1 s, while death at 6 months was predicted by having more than one acute co-morbidity, non-cardiovascular chronic co-morbidity and Activities of Daily Living score.
Conclusions
Chronic and acute co-morbidities are common in COPD patients with AHRF needing NIPPV and their presence influences short and longer-term outcome.
Similar content being viewed by others
Introduction
Prospective randomised controlled studies have shown that the addition of non-invasive positive pressure ventilation (NIPPV) to standard treatment reduces the need for endotracheal intubation, lowers hospital mortality and shortens length of stay in selected patients with chronic obstructive pulmonary disease (COPD) who have acute hypercapnic respiratory failure (AHRF) [1, 2, 3, 4, 5]. Nevertheless, NIPPV is not successful in all cases, with a reported failure rate of 7–50% [6]. Therefore it is important to be able to identify the patients in whom NIPPV is more likely to fail, especially if it is implemented in a respiratory ward. A COPD patient with a high likelihood of failing and for whom invasive ventilation would be appropriate is best managed in the intensive care unit (ICU) where intubation is readily available [6]. Several clinical and physiological respiratory parameters have been shown to predict NIPPV failure [7, 8, 9].
Patients with severe pulmonary dysfunction often suffer from coexistent pathologies and are likely to develop extra-pulmonary complications [10, 11, 12, 13]. A recent Dutch survey reported that 73% of stable COPD patients [14] have at least one chronic non-respiratory co-morbidity (C-NRC) and this is an independent predictor of hospitalisation for exacerbations [15]. C-NRC, usually cardiovascular, were found in 42–97% of patients with COPD exacerbation requiring hospitalisation [13, 16, 17, 18]. C-NRC impacts on short [10, 13, 16] and long-term survival [11, 18] of COPD patients hospitalised with acute decompensation. Furthermore, the development of acute non-respiratory co-morbidity (A-NRC) complicating AHRF may adversely affect hospital [10, 12, 19, 20] and 6-month [19] outcome of severe COPD exacerbations. The effect of non-respiratory co-morbidities upon short and long-term outcome with NIPPV for COPD exacerbation has not been assessed in previous studies [7, 8, 9].
We performed this study [21] in COPD patients with AHRF needing NIPPV to (a) determine the prevalence and the type of C-NRC/A-NRC, defined according to strict criteria, and (b) clarify whether hospital and 6-month outcome are independently affected by C-NRC and/or A-NRC.
Material and methods
Patients
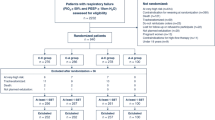
The study protocol was approved by the local ethics committee of our hospital and was performed in accordance with the ethical standards of the 1964 Declaration of Helsinki. Informed consent was obtained in all cases from either the patient or next kin. All 625 patients admitted to our 25-bed respiratory ward in Arezzo between January 1996 and January 2001 for acute decompensation of COPD were considered for the study. Medical treatment consisted of controlled oxygen therapy; salbutamol and ipratropium bromide via aerosol every 4 h; intravenous aminophylline, corticosteroids, antibiotics; cardiovascular drugs and therapy for coexistent C-NRC if necessary. Patients were enrolled in the study if despite this treatment they fulfilled all the following criteria for NIPPV: (a) pH lower than 7.33 or PaCO2 higher than 55 mmHg; (b) PaO2/FIO2 ratio lower than 250; (c) dyspnoea at rest with respiratory rate higher than 25 breaths/min; and (d) use of accessory respiratory muscles or paradoxical abdominal breathing. Exclusion criteria were: (a) refusal of NIPPV; (b) facial deformity sufficient to affect mask fitting; (c) severe encephalopathy unrelated to hypoxaemia and/or hypercapnia; (d) overt gastro-intestinal bleeding; (e) upper airway obstruction; (f) acute ischaemic heart disease; and (g) need for urgent intubation due to cardiac or respiratory arrest, prolonged respiratory pauses and psychomotor agitation requiring sedation [3]. Most of the 625 admitted patients (n=466, 75%) improved quickly with medical therapy. Among the remaining 159 eligible patients with AHRF, 26 (16.4%) were excluded because of the need for urgent intubation and 13 (8.2%) because of refusal of NIPPV. The remaining 120 patients were enrolled in the study 6.25 h (2–47.75) after admission to our ward. Tables 1 and 2 show, respectively, the “historic” and “acute” features of the population.
Non-invasive ventilation
All patients entered in the study were managed in the three-bed respiratory monitoring unit in our respiratory ward [22]. All nurses had attended a 10-h training course on NIPPV in the preceding 2 years; they were kept up to date by means of a yearly 4-h course. Mean nurse:patient ratio was 1:9. During the earliest phase of NIPPV one pulmonologist was continuously present at the patient’s bedside; later the patient was closely looked after by one nurse with one pulmonologist available on call. Electrocardiography, transcutaneous oxygen saturation and non-invasive blood pressure were monitored continuously. Intubation and transfer to ICU was promptly available in the case of NIPPV failure.
NIPPV was delivered by means of Bi-PAP Vision in S/T mode via a well fitting facial mask (Respironics, Murrysville, Pa., USA). A plateau exhalation valve was applied to the circuit to minimise CO2 rebreathing [23]. Inspiratory positive airway pressure (IPAP) was initially set at 10 cmH2O and then gradually increased to a maximum of 25 cmH2O depending on clinical and blood gas response and on the patient’s tolerance. Expiratory positive airway pressure was always set at 5 cmH2O and back-up respiratory rate at 16 breaths/min. FIO2 was adjusted to maintain oxygen saturation at 90% or higher. Patients were encouraged to use NIPPV for as long as tolerated, aiming for at least 6–8 h per day split in three periods; NIPPV was discontinued when all the following criteria were maintained for longer than 24 h while breathing oxygen: pH higher than 7.35, PaO2/FIO2 ratio above 25 and respiratory rate below 25 breaths/min.
Criteria for endotracheal intubation
NIPPV was considered to have failed if at least one of the following criteria for intubation [3] was satisfied: (a) cardiac arrest; (b) respiratory arrest or gasping or prolonged respiratory pauses; (c) psychomotor agitation requiring sedation; (d) severe difficulty clearing bronchial secretions; and (e) worsening of gas exchange (at least two of the following: pH less than 7.30 and less than 0.02 with respect to baseline, PaO2/FIO2 ratio less than 150 and less than 20 with respect to baseline; PaCO2 greater than 20% with respect to baseline) and/or level of consciousness (Kelly and Matthay [24] score above 1 with respect to baseline) during NIPPV for at least 6 h with the best ventilator setting. In the case of refusal of invasive ventilation by the patient and/or relatives family NIPPV failure was considered when the a priori criteria for intubation were met.
Measurements
The following demographic, clinical and functional data were collected when the patient was stable (within 6 months from admission): (a) age and sex; (b) forced expiratory volume in 1 s (FEV1) and arterial blood gases while breathing room air; (c) body mass index; (d) chronic cor pulmonale [18]; (e) functional limitation evaluated with Activities of Daily Living (ADL) score [19]; (f) use of long-term oxygen therapy (LTOT) or home mechanical ventilation (HMV); and (g) prevalence, type (cardiovascular and non) and score of C-NRC [25] (Appendix A). Clinical and functional data collected at admission were: (a) causes of AHRF: community-acquired pneumonia, cardiogenic pulmonary oedema, pulmonary embolism, COPD exacerbation [12]; (b) acute severity of illness assessed by Acute Physiology and Chronic Health Evaluation (APACHE III) score [26]; (c) level of consciousness measured with Kelly-Matthay score; and (d) prevalence and type (cardiovascular and non-cardiovascular) of A-NRC [12] (Appendix B). NIPPV variables were: (a) arterial blood gases while breathing O2 and after a 2-h trial of NIPPV; (b) level of IPAP; and (c) tolerance assessed by a simplified score (1=low, 2=medium, 3=good) [12]. Data collected at 6-month follow-up included: (a) number of readmissions to hospital for further COPD exacerbations and whether mechanical ventilation needed; and (b) mortality calculated from the date of enrolment in the study. Follow-up included a specialist visit with blood gases within 3 weeks after discharge; thereafter all patients were seen every 2 months, unless they deteriorated earlier.
Statistical analysis
Continuous data are expressed as mean (standard deviation) or as median (interquartile range, IQR) if distributed, respectively, normally or not, according to Kolmogorov-Smirnov test (normal distribution: p>0.05); categorical data are presented as percentages. Univariate analysis was first performed for each variable comparing patients successfully treated with NIPPV and those who were not and comparing between subjects who survived and those who had died at 6 months. Continuous variables were analyzed with the two-tailed unpaired t test (parametric data) or Mann-Whitney U test (non-parametric data). Categorical data were compared using the χ2 test or, when appropriate, Fisher’s exact test. A p value less than 0.05 was considered statistically significant.
Due to the limited number of variables that may be entered in the multivariate analysis (one for every ten events), stringent selection criteria were employed [27]. Variables with p value less than 0.01 on univariate analysis were considered eligible for multivariate analysis. To avoid multicollinearity we examined correlations between all eligible variables using Spearman’s test (significance: p<0.01), and those which were likely to be dependent on the others were excluded. Then all selected variables were entered into a stepwise forward logistic regression model (entry criteria: p<0.05); the variables with p value less than 0.05 were considered statistically significant, and their predictive value was reported as the adjusted odds ratio with 95% confidence interval. The goodness-of-fit of the model was assessed by the Hosmer-Lemeshow test [28] (significance: p>0.05). Analyses were performed using version 10.0 of the SPSS statistical software package (SPSS, Chicago Ill., USA).
Results
NIPPV was delivered for a total of 40 h (IQR 22–65.5 h; 1st day 7.5 h, 6–11) over 5.5 days (3–9). NIPPV was well tolerated (compliance score 2, IQR 2–3). IPAP applied was 16 cmH2O (IQR 14–20). NIPPV failed in 22 of 120 cases (18%) 2.5 days (IQR 1–8) after enrolment for: worsening of blood gases (after 6 h of NIPPV: pH 7.24±0.05, PaO2/FIO2 ratio 148±40, PaCO2 98.4±12.2) and/or level of consciousness in 16 (72.7%), psychomotor agitation in 6 (27.3%), secretions retention in 2 (9.1%), cardiac arrest in 2 (9.1%) and gasping in 1 (4.5%); more than one condition caused NIPPV failure in five patients. Only seven subjects were intubated as the others refused intubation and died. Four intubated patients died after 31.5 days (IQR 24.5–55.8) of ICU stay for septic shock (n=2), multiple organ dysfunction syndrome (n=1) or cardiac arrest (n=1). Only three intubated patients were discharged alive (Fig. 1). Overall in-hospital mortality was 15.8% (19/120); length of stay in hospital was 15 days (IQR 11–21). At discharge LTOT and HMV were prescribed in 20 and 8 patients, respectively.
Prevalence of co-morbidity
The prevalence of C-NRC and A-NRC was 20% (24/120) and 41.7% (50/120), respectively. Cardiovascular disease was the commonest co-morbidity. Cardiogenic pulmonary oedema was not counted as A-NRC if it was a cause of AHRF. Non-cardiovascular disorders were more numerous than cardiovascular among C-NRC; the contrary was found regarding A-NRC (Table 3).
In-hospital NIPPV outcome
In-hospital NIPPV failure was greater in patients with than in those without C-NRC (33.3% vs. 14.6%, p=0.043) or A-NRC (32% vs. 8.6%, p=0.001). The higher the number of A-NRC the greater was the chance of NIPPV failure (A-NRC=0:8.6%; A-NRC=1:22.9%; A-NRC>1:53.3%, p<0.001). The presence of cardiovascular A-NRC was significantly associated with NIPPV failure (32.4% vs. 12.8%, p=0.018). Five variables associated with NIPPV failure on univariate analysis (p<0.01) were eligible for entry in the multivariate analysis. Due to the significant correlations found between ADL score and FEV1 (r=0.337, p<0.001), A-NRC and APACHE III (r=0.320, p<0.001), A-NRC and Kelly-Matthay score (r=0.404, p<0.001), FEV1 and the presence of A-NRC were selected to be entered in the multivariable model and were confirmed to be independent predictors of in-hospital outcome (Hosmer-Lemeshow test: χ2=6.449, p=0.597; Table 4). The same result was obtained when the five variables selected in univariate analysis were introduced in the multivariate analysis. If NIPPV failure was expressed as need of intubation and death instead of only need for intubation, short-term outcome was slightly better (19 failures), but the variables selected by univariate and multivariate analysis were the same.
Longer-term NIPPV outcome
All but one of the recruited patients were followed up at 6 months. Of the 100 patients discharged alive from the hospital 49 were then re-admitted for 80 further COPD decompensations, with a mortality of 17.5% (14/80). NIPPV was required in 51 of 80 readmissions (63.8%), with a failure rate of 21.6% (11/51). Intubation was performed in 13 of 80 readmissions (in 6 cases after NIPPV failure). Nine patients died at home: 6 for COPD decompensations, 3 suddenly. Overall 6-month mortality was 35.3% (42/119; Fig. 1). In the subgroup of 51 COPD decompensations subjected to repeat NIPPV after discharge, prevalence of C-NRC and A-NRC was respectively 12% and 32%; a non-significant trend towards a higher rate of NIPPV failure was found in patients with than without A-NRC (55.6% vs. 24%, p=0.083).
In the overall studied population the presence of C-NRC was associated with greater 6-month mortality (54.2% vs. 30.5%, p=0.030); a non-significant trend towards a worse longer-term outcome was observed in patients with than without A-NRC (44.9% vs. 28.6%, p=0.067). Patients with more than one A-NRC had a higher 6-month mortality rate than patients with A-NRC≤1 (66.7% vs. 30.8%, p=0.007). The impact of NIPPV outcome upon longer-term survival was not tested as the high proportion of patients died after NIPPV failure without receiving intubation (15/22) may have introduced a bias.
Four variables significantly associated with 6-month outcome on univariate analysis (p<0.01) were eligible to be entered in the multivariate analysis. Despite the significant association of all C-NRC with longer-term survival in univariate analysis only the presence of non-cardiovascular C-NRC reached the statistical cut-off to be selected for a multivariate analysis (71.4% vs. 30.5%, p=0.005). Due to the correlation between ADL score and HMV (r=−0.291, p=0.001) the presence of more than one A-NRC and of non-cardiovascular C-NRC together with ADL score were selected to be entered in the multivariate analysis and were confirmed as independent predictors of 6-month prognosis (Hosmer-Lemeshow test: χ2=2.366, p=0.669; Table 5). The same result was obtained when the four variables selected in univariate analysis were introduced in the multivariate analysis.
Discussion
This is the first study specifically designed to evaluate the prevalence and the impact of both chronic and acute non-respiratory co-morbidities on the prognosis of COPD patients with AHRF undergoing to NIPPV. We found that C-NRC and A-NRC are common in cases of COPD decompensation receiving NIPPV in a respiratory monitoring unit, non-cardiovascular being more common among C-NRC and cardiovascular among A-NRC. In-hospital NIPPV failure and 6-month mortality rates were higher in patients with than in those without co-morbidities; the former were more influenced by cardiovascular A-NRC the latter by non-cardiovascular C-NRC. The presence of A-NRC was an independent predictor of in-hospital NIPPV failure (together with FEV1), while the presence of more than one A-NRC and of non-cardiovascular C-NRC (together with ADL score) independently predicted 6-month mortality.
Prevalence of co-morbidity
The lower prevalence of chronic co-morbidities (20%) found in our population than in other reports in COPD exacerbations (42–97%) [13, 16, 17, 18] could be explained by the restricted criteria used for C-NRC according to the Charlson et al. [25] index. If we consider all the disorders selected by Charlson et al. including those in which the index was lower than 1, the prevalence of C-NRC in our study (64.2%) did not differ from the others. In common with other published data [10, 11, 13, 16, 17], cardiovascular diseases was the most single frequent disorder. We found at least one A-NRC in 41.7% of patients; Confalonieri et al. [13] reported a higher prevalence (62.5%) in a heterogeneous population (COPD: 59.6%), 63% of whom underwent NIPPV. In our experience, cardiovascular dysfunction was the most common complication, as also reported by other authors [3, 12].
In-hospital NIPPV outcome
The rate of NIPPV failure (18%) and of hospital mortality (15.8%) in our study was similar to other published data [6]. The chance of NIPPV failure increased from 14.6% and 8.6% in patients without to 33.3% and 32% in patients with, respectively, C-NRC and A-NRC. Few authors have assessed the effect of chronic and acute co-morbidities on the outcome of COPD with AHRF and their results are difficult to compare with our data because of the differences in the following: criteria used for defining co-morbidities, setting, severity and causes of AHRF and indications for and technique of mechanical ventilation. In series of COPD requiring conventional mechanical ventilation for an episode of AHRF in ICU chronic left heart failure [29], APACHE II correlated C-NRC and active malignancies [16] were found to be independently associated with hospital mortality. In a study conducted in a respiratory ward [10] a previous myocardial infarction was the only C-NRC significantly associated with in-hospital mortality on univariate analysis; atrial fibrillation and ventricular arrhythmias on admission were independent predictors of hospital mortality on multivariate analysis, together with an alveolar-arterial difference in PO2 greater than 41 mmHg and age. Similarly, we found that the presence of cardiovascular A-NRC was significantly correlated with NIPPV failure. In a study conducted in two respiratory high-dependency units on 137 COPD patients with AHRF [12], the presence on admission of at least one A-NRC together with a lower pH and ADL score were the strongest predictors of late failure after an initial positive response to NIPPV.
Longer-term NIPPV outcome
The 6-month mortality of 35.3% in our series was slightly higher than that reported (up to 30%) by other investigators of COPD non-invasively ventilated for AHRF and is likely to be explained by differences in the severity of illness [9, 30, 31, 32]. The proportion of deaths at 6 months increased from 30.5% and 30.8% in patients without to 54.2% and 66.7% in patient with, respectively, C-NRC and more than one A-NRC. In the largest series of 1,016 COPD patients hospitalised for acute exacerbation and managed without ventilatory support in 65.7% of cases the authors found no clear association between the number of APACHE III correlated C-NRC and 6-month mortality [18]. Conversely, a significant effect of C-NRC on long-term outcome was observed by Antonelli Incalzi et al. [11] in their retrospective study of 270 COPD exacerbations (16% requiring mechanical ventilation); chronic renal failure was the strongest independent predictor of mortality, together with cor pulmonale, FEV1 less than 590 ml, ischaemic heart disease and age. Similarly, we found that the presence of non-cardiovascular C-NRC was independently correlated with longer-term outcome.
Important limitations in our study must be underlined. Firstly, regarding C-NRC we used the Charslon et al. [25] index, which has prognostic implication in geriatric out-patients with chronic diseases but was not calibrated for acute COPD decompensations. Moreover, we evaluated prevalence and prognostic effect of each A-NRC independently of its potentially different weight. However, to our knowledge, an index taking in account both the number and the seriousness of each acute disorder is lacking. Secondly, the limited size of our series may have underestimated the effect of co-morbidities taken singularly; however, we demonstrated the significant impact of cardiovascular A-NRC and non-cardiovascular C-NRC upon short and longer-term outcome, respectively. Thirdly, our models based on the stepwise logistic regression need to be tested prospectively in a similar population of COPD with AHRF. However, the significant Hosmer-Lemeshow goodness-of-fit value indicates no large difference between observed and expected rates of NIPPV failure and longer-term mortality.
In conclusion, this is the first study designed specifically to evaluate the effect of non-respiratory co-morbidities upon short and longer-term outcome with NIPPV for acute COPD exacerbation. We demonstrated that co-morbidities, especially cardiovascular, are common in COPD with AHRF needing NIPPV and are likely to affect hospital and 6-month outcome. When selecting COPD patients for acute NIPPV, physicians should consider not just lung function and respiratory severity scores but also non-respiratory variables.
References
Bott J, Carroll MP, Conway JH, Keilty SEJ, Ward EM, Brown AM, Paul EA, Elliott MW, Godfrey RC, Wedzicha JA, Moxham J (1993) Randomised controlled trial of nasal ventilation in acute ventilatory failure due to chronic obstructive airways disease. Lancet 341:1555–1557
Kramer N, Meyer J, Meharg J, Cece RD, Hill NS (1995) Randomised, prospective trial of noninvasive positive pressure ventilation in acute respiratory failure. Am J Respir Crit Care Med 151:1799–1806
Brochard L, Mancebo J, Wysocki M, Lofaso F, Conti G, Rauss A, Simonneau G, Benito S, Gasparetto A, Lemaire F, Isabey D, Harf A (1995) Noninvasive ventilation for acute exacerbations of chronic obstructive pulmonary disease. N Engl J Med 333:817–822
Celikel T, Sungur M, Ceyhan B, Karakurt S (1998) Comparison of noninvasive positive pressure ventilation with standard medical therapy in hypercapnic acute respiratory failure. Chest 114:1636–1642
Plant PK, Owen JL, Elliott MW (2000) Early use of non-invasive ventilation for acute exacerbations of chronic obstructive pulmonary disease on general respiratory wards. A multicenter randomised controlled trial. Lancet 355:1931–1935
Lightowler JVJ, Elliott MW (2000) Predicting the outcome from NIV for acute exacerbations of COPD. Thorax 55:815–816
Ambrosino N, Foglio K, Rubini F, Clini E, Nava S, Vitacca M (1995) Non-invasive mechanical ventilation in acute respiratory failure due to chronic obstructive pulmonary disease: correlates for success. Thorax 50:750–757
Antón A, Güel R, Gómez J, Serrano J, Castellano A, Carrasco JL, Sanchis J (2000) Predicting the results of noninvasive ventilation in severe acute exacerbations of patients with chronic airflow limitation. Chest 117:823–833
Plant PK, Owen JL, Elliott MW (2001) Non-invasive ventilation in acute exacerbations of chronic obstructive pulmonary disease: long term survival and predictors of in-hospital outcome. Thorax 56:708–712
Fuso L, Antonelli Incalzi R, Pistelli R, Muzzolon R, Valente S, Pagliari G, Gliozzi F, Ciappi G (1995) Predicting mortality of patients hospitalized for acutely exacerbated chronic obstructive pulmonary disease. Am J Med 98:272–277
Antonelli Incalzi R, Fuso L, De Rosa M, Forastiere F, Rapiti E, Nardecchia B, Pistelli R (1997) Co-morbidity contributes to predict mortality of patients with chronic obstructive pulmonary disease. Eur Respir J 10:2794–2800
Moretti M, Cilione C, Tampieri A, Fracchia C, Marchioni A, Nava S (2000) Incidence and causes of non-invasive mechanical ventilation failure after initial success. Thorax 55:819–825
Confalonieri M, Gorini M, Ambrosino N, Mollica C, Corrado A (2001) Respiratory intensive care units in Italy: a national census and prospective cohort study. Thorax 5:373–378
Manen JG van, Bindels PJE, IJzermans CJ, van der Zee JS, Bottema BJAM, Schadé E (2001) Prevalence of comorbidity in patients with a chronic airways obstruction and controls over the age of 40. J Clin Epidemiol 54:287–293
Miravitlles M, Guerrero T, Mayordomo C, Sànchez-Agudo L, Nicolau F, Segù JL (2000) Factors associated with increased risk of exacerbation and hospital admission in a cohort of ambulatory COPD patients: a multiple logistic regression analysis. Respiration 67:495–501
Nevins ML, Epstein SK (2001) Predictors of outcome for patients with COPD requiring invasive mechanical ventilation. Chest 119:1840–1849
Dewan N, Rafique S, Kamwar B, Satpathy H, Ryschon K, Tillotson GS, Niederman MS (2000) Acute exacerbation of COPD. Factors associated with poor treatment outcome. Chest 117:662–671
Connors AF, Dawson NV, Thomas C, Harrell FE, Desbiens N, Fulkerson WJ, Kussin P, Bellamy P, Goldman L, Knaus WA (1996) Outcomes following acute exacerbation of severe chronic obstructive pulmonary disease. Am J Respir Crit Care Med 154:959–967
Seneff MG, Wagner DP, Wagner RP, Zimmerman JE, Knaus WA (1995) Hospital and 1-year survival of patients admitted to intensive care units with acute exacerbation of chronic obstructive pulmonary disease. JAMA 274:1852–1857
Afessa B, Morales IJ, Scanlon PD, Peters SG (2002) Prognostic factors, clinical course, and hospital outcome of patients with chronic obstructive pulmonary disease admitted to an intensive care unit for acute respiratory failure. Crit Care Med 30:1610–1615
Scala R, Bartolucci S, Archinucci I, Coniglio G, Naldi M, Rossi M (2002) Non-respiratory comorbidity and prognosis of non invasive positive pressure mechanical ventilation in acute hypercapnic respiratory failure due to COPD exacerbations. Eur Respir J 20 [Suppl 38]:281
European Respiratory Society Task Force on epidemiology of respiratory intermediate care in Europe (2002) Respiratory intermediate care units: a European survey. Eur Respir J 20:1343–1350
Ferguson GT, Gilmartin M (1995) CO2 rebreathing during BiPAP ventilatory assistance. Am J Respir Crit Care Med 151:1126–1135
Kelly BJ, Matthay HD (1993) Prevalence and severity of neurological dysfunction in critically ill patients. Influence on need for continued mechanical ventilation. Chest 104:1818–1824
Charlson ME, Pompei P, Ales KL, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40:373–383
Knaus WA, Wagner DP, Draper EA, Zimmerman JE, Bergner M, Bastos PG, Sirio CA, Murphy DJ, Lotring T, Damiano A, Harrell FE (1991) The APACHE III prognostic system: risk prediction of hospital mortality for critically ill hospitalised adults. Chest 100:1619–1936
Katz MH. Multivariable analysis: a primer for readers of medical research (2003) Ann Intern Med 138:644–650
Hosmer DW, Lemeshow S (1989) Applied logistic regression. Wiley, New York
Menzies R, Gibbons W, Goldberg P (1989) Determinants of weaning and survival among patients with COPD who require mechanical ventilation for acute respiratory failure. Chest 95:398–405
Confalonieri M, Parigi P, Scartabellati A, Aiolfi S, Scorsetti S, Nava S, Gandola L (1996) Noninvasive mechanical ventilation improves the immediate and long-term outcome of COPD patients with acute respiratory failure. Eur Respir J 9:422–430
Bardi G, Pierotello R, Desideri M, Valdisserri L, Bottai M, Palla A (2000) Nasal ventilation in COPD exacerbations: early and late results of a prospective, controlled study. Eur Respir J 15:98–104
Vitacca M, Clini E, Rubini F, Nava S, Foglio K, Ambrosino N (1996) Non-invasive mechanical ventilation in severe chronic obstructive lung disease and acute respiratory failure: short- and long-term prognosis. Intensive Care Med 22:94–100
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix A: types and score of chronic non-respiratory co-morbidities according to the Charlson et al. index [25]
-
0 points
-
Ischaemic heart diseases (excluded myocardial infarction)
-
Systemic hypertension
-
Atrial or ventricular arrhythmia requiring chronic treatment
-
Haemodynamically significant valvular heart diseases
-
Seizures requiring treatment
-
Parkinson’s disease
-
Thyroid dysfunction
-
Mild renal insufficiency (creatinine >2 mg/dl)
-
Inflammatory bowel diseases
-
-
1 point
-
Chronic heart failure
-
Myocardial infarction
-
Peripheral vascular disease
-
Cerebrovascular disease
-
Mild–moderate diabetes mellitus (without retinopathy, neuropathy or nephropathy)
-
Mild liver disease (without portal hypertension or variceal bleeding)
-
Peptic ulcer disease
-
-
2 points
-
Paraplegia
-
Moderate or severe renal insufficiency (creatinine >3 mg/dl or dialysis and transplant)
-
Severe diabetes mellitus (with retinopathy, neuropathy or nephropathy)
-
Solid tumour without metastases
-
-
3 points
-
Moderate-severe liver disease (with portal hypertension and/or variceal bleeding)
-
-
6 points
-
Metastatic solid tumour
-
Appendix B: types of acute non-respiratory co-morbidities [12]
-
Cardiovascular
-
Shock
-
Cardiogenic pulmonary oedema (not counted if cause of AHRF)
-
Atrial or ventricular arrhythmia requiring treatment
-
Systemic hypertensive peak (systolic blood pressure ≥200 mmHg and/or diastolic blood pressure ≥100 mmHg)
-
Cerebrovascular accident
-
-
Renal
-
Renal insufficiency (creatinine >3 mg/dl with previously normal function)
-
-
Gastrointestinal
-
Hepatic dysfunction
-
Jaundice
-
Pancreatitis
-
-
Neurological
-
Kelly and Matthay score ≥4
-
-
Metabolic
-
Hyponatremia <120 mmol/l
-
Hypo-hyper kalaemia (<3 mmol/l or >6 mmol/l)
-
Fasting hyperglycaemia (>200 mg/dl)
-
-
Haematological
-
Anaemia (haemoglobin <9 g/dl)
-
Thrombocytopenia (platelet count <90,000/mm3)
-
Leucopenia (white blood cell count <3,000/mm3)
-
Rights and permissions
About this article
Cite this article
Scala, R., Bartolucci, S., Naldi, M. et al. Co-morbidity and acute decompensations of COPD requiring non-invasive positive-pressure ventilation. Intensive Care Med 30, 1747–1754 (2004). https://doi.org/10.1007/s00134-004-2368-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-004-2368-4