Abstract
Introduction: Prognostic determination of patients in coma after resuscitation from cardiac arrest is both common and difficult. We explored clinical and electrophysiological testing to determine their associations with favorable and poor outcomes.
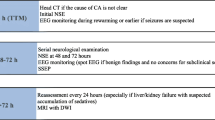
Methods: We studied 75 comatose patients resuscitated from cardiac arrest, excluding those who were brain dead or continuously sedated; none received hypothermia therapy. Clinical examinations were performed on day 1.
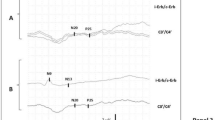
Results: The following proportions recovered awareness: 2 of 18 patients with absent pupillary reflexes; 18 of 57 with preserved pupillary reflexes (p=0.08); 2 of 32 with absent corneal reflexes; 16 of 43 with preserved corneal reflexes (p=0.001); 0 of 15 with absent oculovestibular reflexes; and 8 of 29 with preserved oculovestibular reflexes (p<0.037). Purposeful movements were associated with a high probability of recovery, whereas other categories were unfavorable. Other categories of motor response were associated with an increased proportion of those who died without recovering awareness, but each category had some survivors. Somatosensory evoked potentials (SSEPs) were recorded from 47 patients. One of 21 patients with loss of the N20 component survived, compared with survival of 11 of 26 patients in whom it was present (p=0.003). All 5 patients with preserved N70 responses recovered awareness in a subgroup of 33 patients. Sixteen of 22 subjects with mild electroencephalogram (EEG) abnormalities recovered consciousness, compared with the survival of 3 of 50 patients with malignant EEG patterns (p= 0.0000001). Combining SSEP with EEG findings produced even greater predictive value.
Conclusion: It seems unlikely that any single test will prove to have 100% predictive value for outcome; further studies combining clinical, EEG, and SSEP testing are warranted.
Similar content being viewed by others
References
Bassetti C, Bromio F, Mathis J et al. Early prognosis in coma after cardiac arrest: a prospective clinical, electrophysiological and biochemical study of 60 patients. J Neurol Neurosurg Psychiatry 1996;61:610–615.
Edgren E. Prediction of prognosis following cardiac arrest. Acta Anesthesiol Belg 1988;39:121–126.
Jorgenson EO, Malchow-Moller A. Cerebral prognostic signs during cardiopulmonary resuscitation. Resuscitation 1978;6:217–255.
Levy DE, Bates D, Caronna JJ, et al. Prognosis in nontraumatic coma. Ann Intern Med 1981;94:293–301.
Levy DE, Caronna JJ, Singer BH, et al. Predicting outcome from hypoxic-ischemic coma. J Am Med Assoc 1985;253:1420–1426.
Longstreth WT Jr. Prediction of awakening after out-of-hospital cardiac arrest. N Engl J Med 1983;308:1378–1382.
Pohlmann-Eden B, Dingethal K, Bender H-J, Loelfen W. How reliable is the predictive calue of SEP (somatosensory evoked potentials) patterns in severe brain damage with special regard to the bilateral loss of cortical responses? Intensive Care Med 1997;23:301–308.
Rothstein TL, Thomas EM, Sumi SM. Predicting outcome in hypoxic-ischemic coma. A prospective clinical and electrophysiological study. Electroenceph Clin Neurophysiol 1991;79:101–107.
Zandbergen EG, de Haan RJ, Stoutenbeek CP, Koelman JH, Hijdra A. Systematic review of early prediction of poor outcome in anoxic-ischaemic coma. Lancet. 1998;352:1796–1797.
Rothstein TL. The role of evoked potentials in anoxic-ischemic coma and severe brain trauma. J Clin Neurophysiol 2000;17:486–497.
Logi F, Fischer C, Murri L, Mauguière F. The prognostic value of evoked responses from the primary somatosensory and auditory cortex in comatose patients. Clin Neurophysiol 2003;1615–1627.
Madl C, Grimm G, Kramer L, et al. Early prediction of individual outcome after cardiopulmonary resuscitation. Lancet 1993;341:855–858.
Young GB, McLachlan RS, Kreeft JH, Demelo JD. An electroencephalographic classification for coma. Can J Neurol Sci 1997;24:320–325.
American EEG Society. Guideline Seven: a proposal for standard montages to be used in clinical EEG. American Electroencephalographic Society. 1994;11:30–36.
Ragazzoni A, Ferri R, Di Russo F, et al. Giant somatosensory evoked potentials in different clinical conditions: scalp topography and dipolesource analysis. Electroenceph Clin Neurophysiol 1999;49:81–89.
Desmedt JE, Nguyen TH, Bourget M, Bit-mapped color imaging of human evoked responses with reference to the N20, P22, P27 and N30 somatosensory response. Electroenceph Clin Neurophysiol 1987;68:1–19.
Allison T, McCarthy G, Wood CC, Jones SJ. Potentials evoked in human and monkey cerebral cortex by stimulation of the median nerve. A review of scalp and intracranial montage. Brain 1991;114:2465–2503.
Mima T, Terada K Maekawa M, et al. Somatosensory evoked potentials following proprioceptive stimulation of finger in man. Exp Brain Res 1996;111:233–245.
Deiber MP, Giard MH, Mauguière F. Separate generators with distinct orientations for N20 and P22 somatosensory evoked potentials to finger stimulation. Electroencephalogr Clin Neurophysiol 1986;65:321–334.
Ikeda A, Luders HO, Burgess RC, et al. Generator locations of movement-related potentials with tongue protrusions and vocalizations: subdural recordings in human. Electroencephalog Clin Neurophysiol 1995;96:310–328.
Mauguière F, Allison T, Babiloni C, et al. Somatosensory evoked potentials. The International Federation of Clinical Neurophysiology. Electroencephalogr Clin Neurophysiol 1999;52(Suppl):79–90.
Bernard SA, Gray TW, Buist MD, et al. Treatment of comatose survivors of out-of-hospital cardiac arrest with induced hypothermia. N Eng J Med 2002;346:557–563.
Hypothermia after Cardiac Arrest Study Group. Mild hypothermia to improve the neurologic outcome after cardiac arrest. N Eng J Med 2002;346:549–556.
Deiber MP, Giard MH, Mauguière F. Separate generators with distinct orientations for N20 and P22 somatosensory evoked potentials to finger stimulation. Electroencephagr Clin Neurophysiol 1986;65:321–334
Buchner H, Gobbele R, Pollit D. Radermacher L. Evaluation of the functional state of the somato-motor system using SEP and interfering stimuli. Electroencephalogr Clin Neurophysiol 1996;46(Suppl):351–362.
Barba C, Frot M, Valeriani M, Tonali P, Mauguière F. Distinct fronto-central N60 and supra-sylvian N70 middle-latency components of the median nerve SEPs as assessed by scalp topographic analysis, dipolar source modelling and depth recordings. Clin Neurophysiol 2002;113:981–992.
Young GB. The EEG in coma. J Clin Neurophysiol 2000;17:473–485.
Geocadin RG, Ghodadra R, Kimura T, et al. A novel quantitative EEG injury measure of global cerebral ischemia. Clin Neurophysiol 2000;111:1779–1787.
Young GB, Blume WT, Campbell VM, et al. Alpha, theta and alpha-theta coma: a clinical outcome study utilizing serial recordings. Electroencephalogr Clin Neurophysiol 1994;91:93–99.
Young GB; Kreeft JH; McLachlan RS; Demelo J. EEG and clinical associations with mortality in comatose patients in a general intensive care unit. J Clin Neurophysiol 1999;16:354–360.
Bassetti C, Bomio F, Mathis J, Hess CW. Early prognosis in coma after cardiac arrest: a prospective clinical, electrophysiological, and biochemical study of 60 patients. J Neurol Neurosurg Psychiatry 1996;61:610–615.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Young, G.B., Doig, G. & Ragazzoni, A. Anoxic-ischemic encephalopathy. Neurocrit Care 2, 159–164 (2005). https://doi.org/10.1385/NCC:2:2:159
Issue Date:
DOI: https://doi.org/10.1385/NCC:2:2:159