-
PDF
- Split View
-
Views
-
Cite
Cite
Hong Euy Lim, Hui-Nam Pak, Jeong Cheon Ahn, Woo Hyuk Song, Young-Hoon Kim, Torsade de pointes induced by short-term oral amiodarone therapy, EP Europace, Volume 8, Issue 12, December 2006, Pages 1051–1053, https://doi.org/10.1093/europace/eul118
Close - Share Icon Share
Abstract
Although amiodarone appears to have few pro-arrhythmic effects, torsade de pointes (TdP) has been observed during long-term drug administration, usually in conjunction with electrolyte disturbances, a change in drug dosage, or concomitant drug therapy. We report two cases of amiodarone-induced TdP shortly after administration of a low dose of oral amiodarone, in the absence of predisposing factors.
Introduction
Amiodarone is a well-known class III antiarrhythmic agent. Despite considerable lengthening of the QTc interval and a substantial degree of bradycardia, the drug exhibits a remarkably low frequency of pro-arrhythmic events and an incidence of torsade de pointes (TdP) of <1.0%, a fact mainly attributed to the increased homogeneity of repolarization in contiguous myocardial cells and the lack of calcium-dependent early after-depolarizations.1
However, it is possible that TdP may be provoked in patients on long-term amiodarone therapy, usually in conjunction with electrolyte imbalances, increases in dosage, or by administration of concomitant drugs that prolong the QT interval.2,3
We describe here two cases of TdP induced by short-term low-dose oral amiodarone therapy for the treatment of atrial fibrillation (AF) in the absence of any other predisposing factors.
Case presentations
Case 1
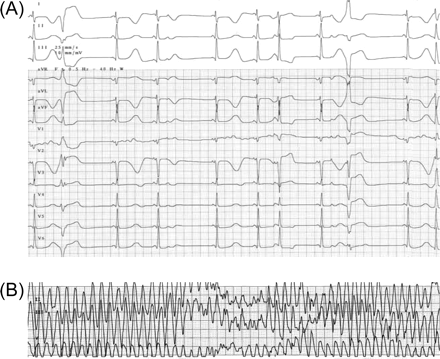
A 40-year-old woman presented with palpitations and severe dizziness. Upon admission, an electrocardiogram (ECG) revealed AF with a rapid ventricular rate of 227 bpm (Figure 1A). Echocardiography showed global wall motion abnormalities of the left ventricle (LV) with an ejection fraction (EF) of 20%. Since the patient had a seriously compromised clinical status, we attempted electrical cardioversion after excluding the presence of a left atrial thrombus by transoesophageal echocardiogram. Following cardioversion, an ECG demonstrated sinus bradycardia (44 bpm) with a QTc interval of 449 ms, accompanied by non-conducted premature atrial complexes (Figure 1B). The patient was considered to be at high risk for recurrence of AF, and oral amiodarone was prescribed at a dose of 200 mg/day with no loading dose because of the bradycardia and hypotension. On the fourth day of treatment, the patient became unconscious and the ECG disclosed a markedly prolonged QTc interval of 533 ms with T-wave alternans (Figure 1C). ECG monitoring revealed frequent premature ventricular complexes with a short coupling interval that was followed by TdP (Figure 2). After defibrillation to restore sinus rhythm, intravenous isoprenaline and magnesium sulphate were administered and the TdP did not recur. At that time, serum electrolytes were within normal limits. The QTc interval gradually decreased to its normal value of 428 ms (Figure 1D) after discontinuation of amiodarone.
(A) Admission ECG revealing AF with a rapid ventricular rate of 227 bpm (B) Following cardioversion, an ECG revealed sinus bradycardia (44 bpm) with a QTc interval at the upper limit of normal for a woman (449 ms), accompanied by non-conducted premature atrial complexes. (C) After receiving oral amiodarone (200 mg/day for 4 days), an ECG showed a markedly prolonged QTc interval of 533 ms with T-wave alternans. (D) The QTc interval normalized to 428 ms after ceasing the oral amiodarone.
On the fourth day of amiodarone treatment, the ECG demonstrated QTc prolongation and frequent premature ventricular complexes with a short coupling interval (A), followed by the occurrence of TdP (B).
Case 2
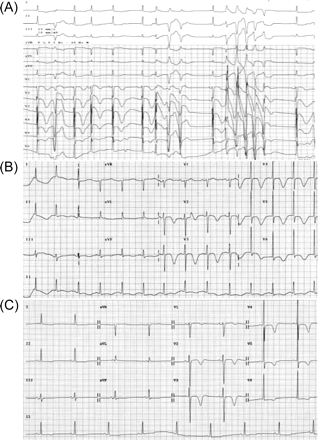
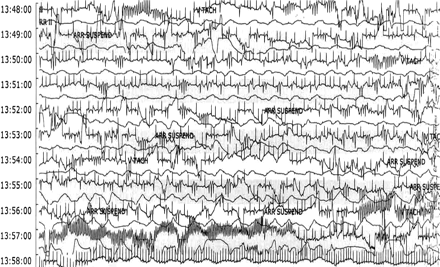
An 85-year-old woman with mild coronary artery disease, hypertension, and dilated cardiomyopathy presented to the emergency room with palpitations and pre-syncope. Her 12-lead ECG revealed a non-sustained, polymorphic ventricular tachycardia with a markedly prolonged QTc interval (528 ms) and broad T-wave inversion (Figure 3A and B). Five days earlier, she had been started on amiodarone (200 mg/day) without a loading dose for paroxysmal AF. Other medications included aspirin 100 mg/day, amlodipine 5 mg/day and candesartan 16 mg/day. Echocardiography revealed mild LV dilatation (57 mm) with an EF of 40%. Laboratory test results were normal. Continuous ECG monitoring showed self-limiting TdP, following frequent ventricular ectopic beats (Figure 4). Intravenous isoprenaline was administered with no relapse of the TdP. After cessation of the oral amiodarone, the QTc interval gradually decreased over the next 7 days. The ECG disclosed normal sinus rhythm with a QTc interval of 446 ms and negative T-waves in all precordial leads (Figure 3C).
(A) Initial ECG revealed non-sustained polymorphic ventricular tachycardia with R on T phenomenon. (B) After administration of isoprenaline, the ECG showed a markedly prolonged QTc interval (528 ms) with broad T-wave inversion and without recurrence of TdP. (C). Note that the QTc (446 ms) was within the normal limit on the seventh day after discontinuation of amiodarone.
Continuous ECG monitoring showing self-limiting TdP following frequent premature ventricular complexes and non-sustained polymorphic ventricular tachycardia.
Discussion
Amiodarone is a commonly prescribed potent antiarrhythmic agent with fewer negative inotropic effects than other antiarrhythmic drugs used for various cardiac conditions.4 Also, compared with other antiarrhythmic agents, it appears to have fewer pro-arrhythmic effects.
The reason for the low incidence of pro-arrhythmia with amiodarone remains unclear, but is probably multifactorial. Although the increase in action potential duration is caused by blocking the delayed rectifier current Ik, amiodarone can also block the fast sodium current and the slow inward calcium current that is mediated through the L-calcium channels.5 The calcium-channel blocking effect may be an important factor with respect to the low incidence of amiodarone-associated TdP. In addition, amiodarone produces a much shorter prolongation of repolarization in Purkinje fibres than in ventricular muscle, so it does not increase QT dispersion in the myocardium.6 However, in certain conditions, including electrolyte disorders and concomitant drugs that prolonged QT interval, amiodarone may also produce TdP.2,3
Drug-induced excessive QT prolongation most commonly occurs in patients with subclinical mutations in one of the genes responsible for the congenital long QT syndrome.7 These patients are susceptible to developing TdP if given a drug that blocks potassium channels. The percentage of carriers with genetic defects in the acquired long QT syndromes is still unknown. Although our patients had no family history of congenital long QT syndrome or sudden cardiac death caused by ventricular tachyarrhythmias, and their ECGs revealed normal values of QTc interval after ceasing oral amiodarone, they could belong to the susceptible group of patients who have silent mutations in the function of their ion channels.
These two cases of TdP occurring early in oral amiodarone therapy at a low dose (200 mg/day), and in the absence of other predisposing factors, emphasize the importance of careful patient monitoring during amiodarone therapy, even with short-term use.