Abstract
Purpose: A pharmacokinetic study in children to determine plasma flumazenil concentrations after the intranasal administration of 40 µg·kg−1.
Methods: Following institutional approval and informed written consent, II ASA physical status I–II patients, aged two to six years, undergoing general anesthesia for dental surgery were recruited. After induction, 40 µg·kg−1 flumazenil Anexate®, Roche, 0.1 mg·mL−1 (0.4 mL·kg−1)) were administered via a syringe as drops, prior to nasal intubation. Venous plasma samples were drawn prior to administration of flumazenil (t=0), and then at 2, 4, 6, 8, 10, 15, 20, 30, 40, 60, and 120 min thereafter. The plasma samples were immediately processed by the onsite laboratory and then stored at −70°C, before batch analysis via high performance liquid chromatography assay. Pharmacokinetic data calculations were performed using WinNonLin software (Scientific Consulting Inc.).
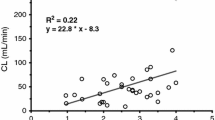
Results: Eleven patients were studied, but data for one patient were discarded due to insufficient sampling. The median age was 4.3 yr (range 3 to 6), with a median weight of 18.9 kg (range 14.9 to 22.2). There were seven boys and three girls. Mean Cmax was 67.8 ng·mL−1 (SD 41.9), with Tmax at two minutes. The calculated half-life was 122 min (SD 99).
Conclusion: The mean plasma concentrations of flumazenil attained were similar to those reported after intravenous administration, and may be sufficient to antagonize the side-effects of benzodiazepines. This route of administration may be useful when the intravenous route is not readily available.
Résumé
Objectif: Déterminer, par une étude pharmacocinétique chez des enfants, la concentration plasmatique de flumazénil après l’administration intranasale de 40 µg·kg−1.
Méthode: Après avoir obtenu les autorisations écrites de l’institution et des parents, on a recruté II patients, d’état physique ASA I–II, âgés de deux à six ans, qui devaient subir une intervention dentaire sous anesthésie générale. À la suite de l’induction, 40µg·kg−1 de flumazénil (Anexate®, Roche, 0,1 mg·mL−1 (0,4 mL·kg−1)) ont été administrés en gouttelettes au moyen d’une seringue avant l’intubation nasale. Des échantillons plasmatiques veineux ont été pris avant l’administration de flumazénil (t=0), et ensuite à 2, 4, 6, 8, 10, 15, 20, 30, 40, 60, et 120 min. Les échantillons plasmatiques ont été immédiatement traités au laboratoire sur place et conservés à −70 °C, avant l’analyse du tout par chromatographie à haute performance. Les calculs pharmacocinétiques ont été réalisés avec le logiciel WinNonLin (Scientific Consulting Inc.).
Résultats: Les données d’un seul patient sur II ont été refusées pour échantillons insuffisants. L’âge moyen des en fants, dont sept garçons et trois filles, était de 4,3 ans (limites de 3 à 6), et le poids moyen était de 18,9 kg (limites de 14,9 à 22,2). La Cmax moyenne était de 67,8 ng·mL−1 (écart type de 41,9), et un Tmax à deux minutes. La demi-vie était de 122 min (écart type de 99).
Conclusion: Les concentrations plasmatiques moyennes de flumazénil obtenues ont été similaires à celles qui suivent l’administration intraveineuse et peuvent être suffisantes pour contrer les effets secondaires des benzodiazépines. L’administration par voie nasale peut se révéler utile lorsque la voie intraveineuse n’est pas facilement accessible.
Article PDF
Similar content being viewed by others
References
Malinovsky J-M, Lejus C, Servin F, et al. Plasma concentrations of midazolam after I.V., nasal or rectal administration in children. Br J Anaesth 1993; 70: 617–20.
Malinovsky J-M, Populaire C, Cozian A, Lepage J-Y, Lejus C, Pinaud M. Premedication with midazolam in children. Effect of intranasal, rectal and oral routes on plasma midazolam concentrations. Anaesthesia 1995; 50: 351–4.
Ricou B, Forster A, Brückner A, Chastonay P, Gemperle M. Clinical evaluation of a specific benzodiazepine antagonist (Ro 15-1788). Studies in elderly patients after regional anaesthesia under benzodiazepine sedation. Br J Anaesth 1986; 58: 1005–11.
Klotz U, Kanto J. Pharmacokinetics and clinical use of flumazenil (Ro 15-1788). Clin Pharmacokinet 1988; 14: 1–12.
Amrein R, Hetzel W, Hartmann D, Lorscheid T. Clinical pharmacology of flumazenil. Eur J Anaesthesiol 1988; S2: 65–80.
Perry HE, Shannon MW. Diagnosis and management of opioid- and benzodiazepine-induced comatose overdose in children. Curr Opin Pediatr 1996; 8: 243–7.
Jones RDM, Lawson AD, Andrew LJ, Gunawardene WMS, Bacon-Shone J. Antagonism of the hypnotic effect of midazolam in children: a randomized, double-blind study of placebo and flumazenil administered after midazolam-induced anaesthesia. Br J Anaesth 1991; 66: 660–6.
Richard P, Autret E, Bardol J, et al. The use of flumazenil in a neonate. J Toxicol Clin Toxicol 1991; 29: 137–40.
Walbergh EJ, Wills RJ, Eckhert J. Plasma concentrations of midazolam in children following intranasal administration. Anesthesiology 1991; 74: 233–5.
Vletter AA, Burm AG, Breimer LTM, Spierdijk J. High-performance liquid chromatographic assay to determine midazolam and flumazenil simultaneously in human plasma. J Chromatogr 1990; 530: 177–85.
Van Der Bijl P, Roelofse JA. Disinhibitory reactions to benzodiazepines: a review. J Oral Maxillofac Surg 1991; 49: 519–23.
Bailey PL, Pace NL, Ashburn MA, Moll JWB, East KA, Stanley TH. Frequent hypoxemia and apnea after sedation with midazolam and fentanyl. Anesthesiology 1990; 73: 826–30.
Roelofse JA, Stegmann DH, Hartshorne J, Joubert JJ. Paradoxical reactions to rectal midazolam as premedication in children. Int J Oral Maxillofac Surg 1990; 19: 2–6.
Jones RDM, Chan K, Roulson CJ, Brown AG, Smith ID, Mya GH. Pharmacokinetics of flumazenil and midazolam. Br J Anaesth 1993; 70: 286–92.
Philip BK. Drug reversal: benzodiazepine receptors and antagonists. J Clin Anesth 1993; 5: 46S-51.
Breheny FX. Reversal of midazolam sedation with flumazenil. Crit Care Med 1992; 20: 736–9.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was funded by a grant from the Vancouver Foundation.
Rights and permissions
About this article
Cite this article
Scheepers, L.D., Montgomery, C.J., Kinahan, A.M. et al. Plasma concentrations of flumazenil following intranasal administration in children. Can J Anaesth 47, 120–124 (2000). https://doi.org/10.1007/BF03018846
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03018846